Table of Contents
What is Sulphur Dye?
The dyes are so-called as they contain sulpher linkage but dissolve in Na-sulphide solution which acts as a reducing agent, breaking the sulpher linkage & converting the molecules into simpler components that are soluble in water & substantive towards cellulose. General formula: Dye-S-S-Dye
Chemical Structure:
- When sulfur is heated with P-toluidine, it forms dehydrothio-toluidine.
- The dehydrothio-toluidine can react with another molecule of toluidine process which may be repeated until quite a complex molecule of toluidine a process which may be repeated until quite a complex molecule has been built up:

Properties of Sulpher Dyes:
✔ They contain sulfur linkage within their molecule.
✔ Water Insoluble dyes.
✔ Heat reduction and oxidation of the development in the fiber.
✔ Wet fastness is good.
✔ Light fastness satisfactory.
✔ Wash fastness excellent.
✔ Limited range of color normally black.
✔ Chlorine fastness fair.
✔ Shading is not more than 10%.
✔ Unhygienic for the environment.
✔ Very cheap.
✔ Available in the market.
✔ S-dye makes colloidal solution.
✔ S-dye produces Hydrogen Sulphide to decompose.
Mechanism of Dyeing with S-dye:
▪ Sodium sulfide acts as a reducing agent that breaks the sulfur linkage of dyes & breaks down the molecules into simpler components that can easily penetrate fabric surfaces.
Dye-S-S-Dye + 2[H]
Reducing agentDye-S-H + H-S-Dye
Insoluble Soluble (Leucoform)
▪ The thiols, containing the –SH groups are readily oxidized in the fiber to the original insoluble sulfur dyes by oxidizing agent and give a color with very good wet fastness properly.
Oxidizing Agent
2 Dye-S-H + [O]
Dye-S-S-Dye +H2O Insoluble Sulpher dye
▪ Dyes are negatively ionized and have no Vander Waals force effect on them, salt improves dyeing efficiency by increasing physical force.
Dyeing of Cotton by Sulphur Dyes:
▪ Recipe (Std):
Sulpher dyes: 10-20%
Wetting agent: 1-2%
Sodium sulphide: 10-20%
Soda ash (PH control): 5-10%
Common Salt (Electrolyte): 15-20%
M:L = 1:20
Time: 1 hr
Temp: 700-1000C
▪ Process Sequence: Fabric preparation → Preparation of dye solution → Dyeing (suitable method) → Oxidation → After treatment
▪ Fabric Preparation: Only normal pretreated (Scoring, bleaching) fabric is required. Mercerized cotton goods cause an increased color yield of 30 to 40%.
▪ Dye Reducing Step: The required amount of dye, soda ash, sodium sulfide, and wetting agent are taken in a beaker & little amount of cold water is added to make a paste. In a separate vessel, the rest of the water is boiled & added to the paste to make the stock solution which was further boiled for 5 mins so that the day completely reduced.
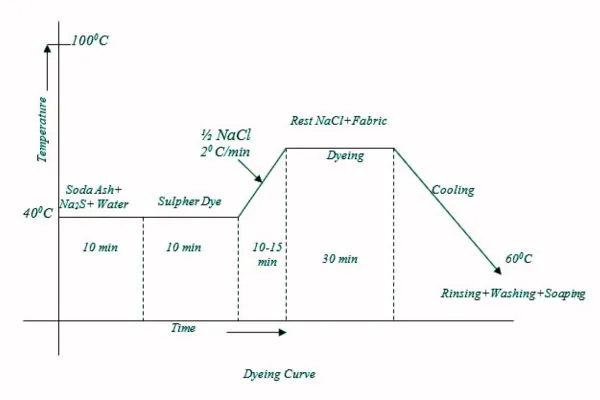
▪ Dyeing: At first dye solution is taken in a dye bath and the fabric is immersed in that solution and then heated for 10-15 min. Then half of the total NaCl solution is added and the temperature is raised up to boil (100oC) and the rest of the salt is added. Dyeing at this temp is continued for 30 min.
▪ Oxidation: After dyeing, the reduced water-soluble form of the dye is oxidized to fix the dyestuff as its insoluble oxidized form. Typical recipe for oxidation
Na-perborate: 0.5-1 g/L OR 1.3 of
Temp: 40-500C
Time: 20 min
▪ After treatment:
- The dyed substance is soaped which makes the color bright & gives permanent shade. b. A treatment with CuSO4 & acetic acid in the presence of sodium or potassium dichromate improves the light fastness. When the dichromate is present, the washing fastness is slightly improved.
CuSO4: 0.5-1%
Na/K-dichromate: 1-1.5 ”
Acetic acid: 1-2% ”
Time: 20-30 min
Temp: 700C
Disadvantages / Defects of Sulphur dyes:
- Brounziness of shades: It is a common defect found in sulpher-dyed textile materials. o Causes:
✔ Excessive delay between lifting of materials out of the dye bath & washing off.
✔ The presence of excessive dyestuff on the material.
✔ Insufficient amount of sodium sulfide in the dye bath.
✔ Exposure of goods to air while dyeing.
✔ Excessive heat.
o Remedies:
✔ Good washing and dilute solution of Na2S (1%) at 30oC OR
✔ A treatment with boiling soap solution or a strong Na2S solution OR
✔ A treatment with a solution containing 10% saponified palm oil at 60 C.
- Tendering: If sulpher-dyed textile materials are stored, a tendering effect is seen on cellulose. o Causes:
✔ Gradual oxidation of sulfur to sulphuric acid on storage.
✔ After treatment with copper salt causes rapid tendering.
✔ The presence of ion as an impurity causes rapid tendering.
✔ The method of oxidation for the reconversion to insoluble form influences tendering.
o Remedies:
✔ Treatment of dyed materials with a little sodium acetate so that H2So4 may be converted into harmless acetic acid.
Advantage of S-dye / Why S-dyes are so popular in producing black shade:
✔ Low cost
✔ Fair to good light fastness.
✔ High wash fastness.
✔ Easy to apply.
✔ Low energy required.
✔ Chemical resistance is moderate to good.
✔ Wet fastness good
✔ Chlorine fastness fair.
Uses of S-dye:
✔ Used to dye black umbrella cloth.
✔ Used for lining boats.
✔ Used to dye rubber materials.

Mahedi Hasan working as an Executive (Fabric Marketing) at Pengnuo Group. Graduated with B.Sc. in Textile Engineering. Before was a Top Rated content writer at Upwork, and Level 02 Seller at Fiverr, Level 02 Publisher at Ezoic. Very passionate about content writing, SEO practice, and fashion website designing. Highly Experienced fashion writer for the last 4+ years. Have extensive 7 years of experience in the wholesale clothing business.
